The volume to mass calculator is a tool that will help you convert volume to mass or mass to volume. It includes dozens of items and their densities divided into six categories to help you find your result in seconds. Do you want to know how to find volume with density and mass? Keep reading and find out!
Check how water density changes with temperature and pressure with our water density calculator.
How to find volume with density and mass?
To convert something's mass to volume or vice versa, you need to know its density. What is density? It's mass-to-volume ratio — a physical property of every material. For example, water at 4 °C has a density of 1 kg/l. It means that one liter of water weighs one kilogram. You've probably already guessed what formula will tie these three values together:
density = mass / volume
It's worth knowing that density slightly changes with temperature and pressure. While it doesn't matter in most cases (like cooking), it might be significant if you're building something or doing a science experiment.
So, how do you calculate volume with mass and density? You need to rearrange the formula to:
volume = mass / density
Whenever you use this formula, remember to be concise with units. For example, if you input mass in pounds and volume in gallons, you will get density in pounds per gallon. In our volume to mass calculator, you can input values in any unit you want — our calculator will convert them and make correct calculations.
How to use our volume to mass calculator?
-
First, you need density. Check if the material is on our list or input known density.
-
Second, input volume or mass in the correct unit. Change the unit first before you enter a number.
-
That's it! The volume to mass calculator will find the result in less than a second!
Here are the products and their densities available in our calculator:
- Food:
- Water — 1,000 kg/m3
- Milk — 1,030 kg/m3
- Flour
- Buckwheat — 660 kg/m3
- Barley — 610 kg/m3
- Corn — 550 kg/m3
- Rye — 670 kg/m3
- Wheat — 600 kg/m3
- Soy — 680 kg/m3
- Corn starch — 650 kg/m3
- Potato starch — 720 kg/m3
- Sugar
- Granulated sugar — 845 kg/m3
- Powder sugar — 560 kg/m3
- Brown sugar — 800 kg/m3
- Salt — 1,217 kg/m3
- Honey — 1,420 kg/m3
- Butter — 959 kg/m3
- Oil
- Cooking oil — 880 kg/m3
- Olive oil — 918 kg/m3
- Sunflower oil — 960 kg/m3
- Vegetable oil — 890 kg/m3
- Nuts and Seeds
- Almonds, ground — 440 kg/m3
- Walnuts, hazelnuts, grounded — 520 kg/m3
- Sesame — 640 kg/m3
- Sunflower seeds — 620 kg/m3
- Peanuts, shelled — 690 kg/m3
- Cacao — 520 kg/m3
- Rice (raw) — 850 kg/m3
- Oats — 410 kg/m3
- Jam — 1 330 kg/m3
- Nutella — 1,260 kg/m3
- Maple syrup — 1,320 kg/m3
- Cream 38% fat — 984 kg/m3
- Cream 13% fat — 1,013 kg/m3
- Metals:
- Aluminum — 2,700 kg/m3
- Beryllium — 1,850 kg/m3
- Brass — 8,600 kg/m3
- Copper — 8,940 kg/m3
- Gold — 19,320 kg/m3
- Iron — 7,870 kg/m3
- Lead — 11,340 kg/m3
- Magnesium — 1,740 kg/m3
- Mercury — 13,546 kg/m3
- Nickel — 8,900 kg/m3
- Platinum — 21,450 kg/m3
- Plutonium — 19,840 kg/m3
- Potassium — 860 kg/m3
- Silver — 10,500 kg/m3
- Sodium — 970 kg/m3
- Tin — 7,310 kg/m3
- Titanium — 240 kg/m3
- Uranium — 18,800 kg/m3
- Zinc — 7,000 kg/m3
- Non-metals:
- Concrete — 2,400 kg/m3
- Cork — 240 kg/m3
- Diamond — 3,500 kg/m3
- Ice — 916 kg/m3
- Nylon — 1,150 kg/m3
- Oak — 710 kg/m3
- Pine — 373 kg/m3
- Plastics — 1,175 kg/m3
- Styrofoam — 75 kg/m3
- Wood (typical) — 700 kg/m3
- Gases:
- Air (sea level, 0 °C) — 1.293 kg/m3
- Air (sea level, 20 °C) — 1.205 kg/m3
- Carbon dioxide (sea level, 0 °C) — 1.977 kg/m3
- Carbon dioxide (sea level, 20 °C) — 1.842 kg/m3
- Carbon monoxide (sea level, 0 °C) — 1.250 kg/m3
- Carbon monoxide (sea level, 20 °C) — 1.165 kg/m3
- Hydrogen — 0.0898 kg/m3
- Helium — 0.179 kg/m3
- Methane (sea level, 0 °C) — 0.717 kg/m3
- Methane (sea level, 20 °C) — 0.688 kg/m3
- Nitrogen (sea level, 0 °C) — 1.2506 kg/m3
- Nitrogen (sea level, 20 °C) — 1.165 kg/m3
- Oxygen (sea level, 0 °C) — 1.4290 kg/m3
- Oxygen (sea level, 20 °C) — 1.331 kg/m3
- Propane (sea level, 20 °C) — 1.882 kg/m3
- Water vapor — 0.804 kg/m3
- Liquids:
- Liquid hydrogen (-255 °C) — 70 kg/m3
- Liquid oxygen (-219 °C) — 1,141 kg/m3
- Water (fresh, 4 °C) — 1,000 kg/m3
- Water (salt, 3%) — 1,030 kg/m3
- Astronomy:
- The Universe — 5×10-27 kg/m3
- Interstellar medium — 1×10-19 kg/m3
- The Earth — 5,515 kg/m3
- Earth's inner core — 13,000 kg/m3
- Sun's core (min) — 33,000 kg/m3
- Sun's core (max) — 160,000 kg/m3
- Super-massive black hole — 9×105 kg/m3
- White dwarf star — 2.1×109 kg/m3
- Atomic nuclei — 2.3×1017 kg/m3
- Neutron star — 4.8×1017 kg/m3
- Stellar-mass black hole — 1×1018 kg/m3
🔎 Fun fact: Although the universe consists of many super-dense objects like stars and black holes, it's almost empty.
As you see, we listed all the densities in kilograms per cubic meter. If you want to know the density in any other unit, you can choose the desired product in our calculator and change the density unit to a different one or go to the density converter.
Density-mass-volume triangle
The density-mass-volume triangle is a helpful trick to help you remember the density formula:
density = mass / volume
To draw a density-mass-volume triangle, you have to:
- Draw a triangle;
- Divide it into three parts with two lines; and
- Write down the mass in the top part and density and volume in the bottom parts.
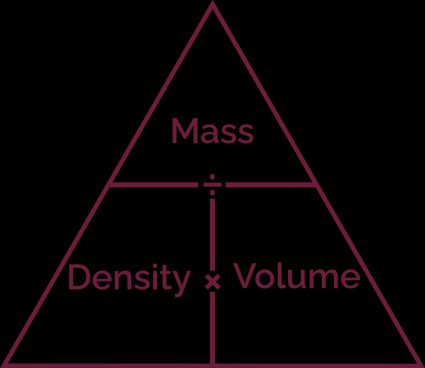
Do you see how the horizontal line looks like a fractional dash? Looking at this triangle, you can deduce that:
- Density equals mass divided by volume;
- Volume equals mass divided by density; and
- Mass equals density times volume.
Now, without trouble, you will always remember how to convert volume to mass. And if you don't know the density – we might have it in our volume to mass calculator! So, feel free to use it whenever you need it!
FAQs
How do I find density with mass and volume?
To find density with mass and volume, you simply need to divide the mass by the volume, as shown in the formula:
density = mass / volume
However, if you wish to simplify the process further, Omni Calculator’s volume to mass calculator is at your disposal.
What’s the density-mass-volume triangle?
The density-mass-volume triangle is a little trick to help you remember the density formula. Picture a triangle divided into three sections, with mass at the top and density and volume at the two bottom sections. Since the horizontal line looks like a fraction symbol, this reminds you that mass / density = volume and mass / volume = density.
How many grams are in a cup of water?
There are 237 grams in a cup of water. To get this value:
-
Use the density formula:
density = mass/volume -
Rearrange the formula as:
mass = density × volume -
Substitute the values into the formula. We assume water has a density of 1 g/ml, and 1 cup is equal to 237 ml.
-
This means that there are 237 grams in a cup of water.
When using this formula, ensure all values are in consistent units. Alternatively, simplify the conversions by using Omni Calculator’s volume to mass calculator.
What’s the volume of 10 grams of air?
The volume is 8299 cm³. To determine the volume of 10 grams of air, use the formula:
volume = mass / density
Assuming air at 20 °C with a density of 0.001205 g/cm³, we can substitute the known values into the formula to find that the volume occupied by 10 grams of air is 8299 cm³.