If you've ever wondered how weather reports are able to calculate the humidity so precisely, our mixing ratio of air calculator can help. Read on to find out how these two quantities are related, how they are affected by increasing the vapor pressure, and how to take the measurement of water vapor in the air! We will also show you how to find the actual mixing ratio, of course!
Bear in mind that this tool calculates the mixing ratio in a physical (or meteorological) sense. If you're interested in chemistry, you may want to check our other mixing ratio calculator instead.
What are the actual and saturation mixing ratios?
The actual mixing ratio is defined as the mass of the water vapor to the mass of dry air. It's similar to the specific humidity, although this quantity is related to the total dry air plus aqueous vapor. It isn't commonly used in daily life, but what about science?
It turns out that the results from the mixing ratio of the air calculator can be used in:
-
Meteorology, to compute the relative humidity and to trace the properties of air masses. Taking it further, this can be used in weather predictions and help you sleep better by ensuring that your well-deserved vacation is unlikely to be disturbed by unfavorable atmospheric conditions.
-
Engineering, for example, in calculations related to air conditioning and transporting the water vapor to control room humidity. You may appreciate it, especially during winter when heaters cause the air to become dry and unpleasant.
-
Microclimate studies, to investigate the infiltration results, which occur when the outside air is accidentally introduced into a building. The actual mixing ratio is a more sensitive way of differentiating various air streams than the relative humidity. For instance, a hot kitchen and a cold Alaskan street may have similar humidity but different mixing ratios.
-
Atmospheric thermodynamics to find the value of the virtual temperature of a parcel of air. This is the temperature dry air would have if it had the same pressure and density as a given sample of moist air.
🔎 Virtual temperature is, in fact, a very real quantity. If you'd like to learn more about it, visit our virtual temperature calculator!
What is a saturation mixing ratio, then? This is the maximum amount of water vapor the air can hold without condensation at a particular temperature and pressure. Understanding that the air is saturated when it can't hold any additional H2O in the gaseous state may help grasp the concept.
How to take a measurement of water vapor in the air?
H2O in its gaseous state influences many things such as cloud formation, precipitation, or the heat index. Therefore, it's only natural that we may want to measure its amount to forecast the weather. You can do this either by using a special device such as a hygrometer or with calculations, for instance, with the help of our mixing ratio of air calculator.
It's worth noticing that although the mixing ratio and absolute humidity both include information about how much water vapor is in the air, they are hardly ever used in daily life. Instead, weather reports tend to focus on the relative humidity, which measures how close the air is to being saturated and therefore is expressed as a percentage.
What is the mixing ratio equation? The mixing ratio of air calculator
Although the mixing ratio equation itself isn't overly complicated, there are a few quantities you'll need to establish along the way. You can find them below as well as how to find the relative humidity:
- Determine the actual vapor pressure, , using the equation:
where:
- is the dew point temperature in Celcius; and
- has the base unit hPa or mb.
If you are wondering what dew point temperature is, check our dew point calculator.
-
Find the station pressure, (also in hPa or mb). Don't be discouraged by its name; you don't actually need to be at a station. Use the air pressure of your location. If you can't measure it directly, the air pressure at altitude calculator may be helpful.
-
How to find the actual mixing ratio, ? It can be obtained using the formula:
- If you choose to go further, you'll need the saturated vapor pressure, , obtained from the equation:
where:
- is the temperature of the air in Celcius.
- Then the saturation mixing ratio, , is found from:
- Time to bring it all together! The relative humidity formula is simple and looks like this:
This air mixing ratio calculator hides the vapor pressure of H2O, but if you want to know its value, you can find it by ticking the option "Show vapor pressures" at the bottom.
Humidity and mixing ratio in meteorology — an example
If you're still wondering why the mixing ratio of the air calculator could be useful to you, here's a practical example. You could have noticed that as it makes you experience the air temperature as higher than it actually is. Wind chill causes a similar (opposite) effect in winter.
Therefore, if you're planning to do some activity, whether it's just a pleasant hike or intensive exercise, it's good to know what will await you, especially if you're concerned with your scores, such as VO2 max.
Let's use data for San Antonio, Texas. On some day in June, the temperature was 81 °F (27.22 °C), with a dew point of 78 °F (25.56 °C) and pressure of 1006 mb. Have you noticed how close the air temperature was to the dew point? Because of that, we may expect that the air was close to being saturated with water vapor. Inputting the numbers into the calculator, we obtain the actual mixing ratio of 20.87 g/kg and a relative humidity of 90.34%, confirming our ansatz.
Phoenix, Arizona, on the contrary, was reported to have an air temperature of 88 °F (31.11 °C), a dew point of 54 °F (12.22 °C), and exactly the same pressure. This resulted in the mixing ratio of 8.92 g/kg, with a relative humidity of 30.55%. So, although it was hotter, you most likely would've found it easier to exercise in Phoenix on that day than in San Antonio.
We've prepared a small mixing ratio chart. It illustrates how the mixing ratio would change depending on the dew point under the conditions in San Antonio, that is, air temperature of 80.6 °F (27 °C) and pressure of 1006 mb:
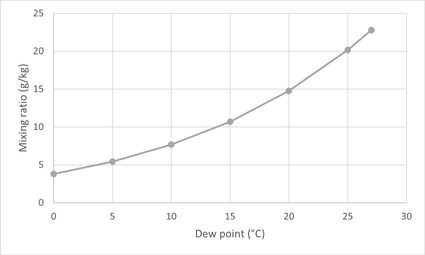
It can also give you an idea of how increasing the vapor pressure would affect the mixing ratio since it depends on the dew point.
You could use a similar approach when considering your summer voyage or preparing for competition!
FAQs
What is the dew point of the air?
The dew point meaning is the temperature at which air is saturated with water vapor. It can be found using the formula:
Dew point = Air temperature - (100 - relative humidity)/5
What's the difference between dew point and humidity?
The dew point is the temperature at which the air becomes saturated with water vapor, whereas relative humidity is the percent of saturation at a given temperature. In other words, the dew point is related to the quantity of moisture in the air and tells us at what temperature the relative humidity would be 100%.
What happens when dew point and air temperature are the same?
When the dew point and air temperature are the same, the air is saturated and the relative humidity is 100%. Water vapor may start to condense and form fog.
How does increasing the vapor pressure affect the mixing ratio?
The mixing ratio is proportional to the actual vapor pressure, so making one quantity larger will yield an increase in another. Because of that, if the dew point rises, so does the vapor pressure and the mixing ratio.